THE GMP Specification is the highest grade of internal fit-out that can be selected for any Cleanroom Solutions project, and is most appropriate for cGMP applications and facilities audited by MHRA and the FDA.
All internal components selected are suitable for high grade Pharmaceutical and clinical environments where attention to detail, quality of finish and reduction of ledges is critical. The essential elements of this internal level of Specification relate to a bespoke panel system with specific connection to flooring and ceiling materials with special consideration to environmental monitoring and 21 CFR Part 11 compliance. Attention to utility integration is also a key part of the GMP Specification, with specially designed raceways and conduits that are embedded within the panel system to eliminate any surface mounted wiring or pipework and containment, also facilitating flush integrated sockets. The GMP Specification adopts best practice throughout and aids compliance to EU GMP Annex 1, ISO 14644-1 and US FS209E, with full qualification process including Design (DQ), Installation (IQ) and Operational (OQ).

Saakvee Make an Enquiry
Got a question for Saakvee Commercial team? Click the button below to get rapid, direct access to Commercial Director Mark Wheeler, Commercial Manager Andrew Cressey, Marketing Manager Joe Shackley and Klensys Cleanroom Consultant Jordan Lucas.
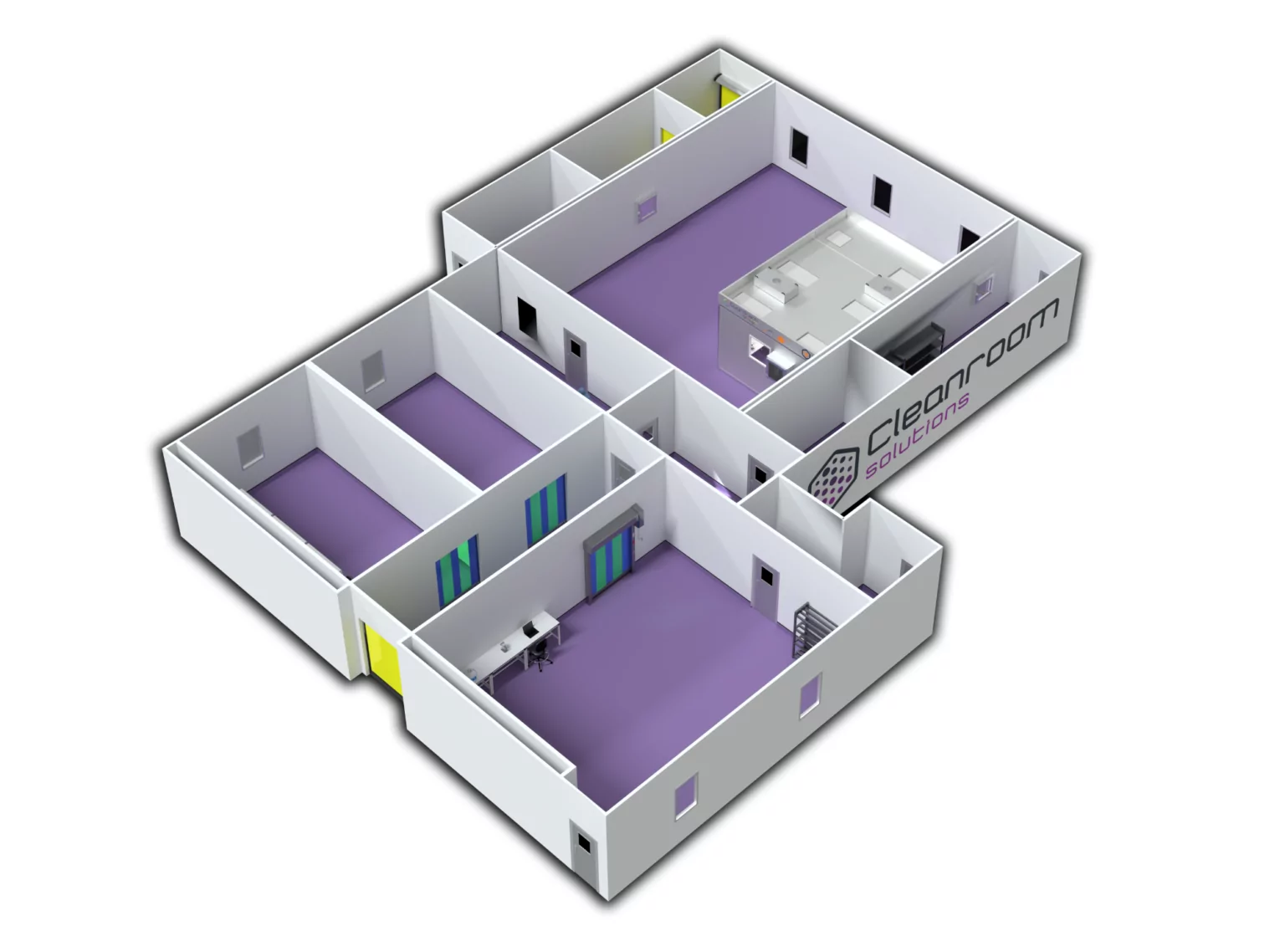
Saakvee CASE STUDY THREE-PHASE PHARMA MANUFACTURING FACILITY
Discover the design innovation, processes and challenges behind an epic three-phase Pharmaceuticals manufacturing facility complete with CNC lab and packaging zone.

Furniture & Equipment Klensys Cleanroom Hardware
Does your cleanroom require a stainless steel furniture fit-out? Maybe it’s some lab-grade Trespa gear you require? Laminar flow units? Biosafety cabinets? Transfer hatches? Whatever it is, Klensys Cleanroom Hardware will have the solution. Browse the range now.

